Percutaneous Stabilization of a Helex Device following Locking-Loop Fracture: A Case Report
ABSTRACT: Though present in up to a quarter of adults a patent foramen ovale (PFO) has been implicated in a diverse range of medical conditions including cryptogenic stroke, migraine and decompression illness. A wide range of devices are available, each with different technical advantages that may favor particular anatomic features. We recently encountered a case of late-locking loop fracture following the use of a Helex septal occluder device (W.L. Gore and Associates, Flagstaff, Arizona) to close a PFO in a diver. This resulted in increased device mobility and continued interatrial shunting. Here we describe the successful stabilization of the original device by transcatheter insertion of a second, less compliant device through the residual atrial shunt. Key words: patent foramen ovale; congenital heart disease; Helex device; decompression illness
_______________________________________________
J INVASIVE CARDIOL 2009;21:E213–E215
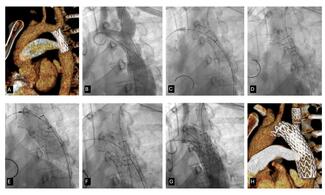
Case Report. A 43 year-old professional diving instructor was referred to the Wessex Adult Congenital Heart Unit for consideration of transcatheter patent foramen ovale (PFO) closure. He had suffered an episode of neurological decompression sickness following a dive and a transesophageal echocardiogram (TEE) demonstrated a PFO with a resting left-to-right shunt which reversed during Valsalva (Figure 1). The PFO was composed of a long tunnel and the associated atrial septal anatomy was complex. The latter involved a relatively wide separation of septum primum and septum secundum, and additionally, the Eustachian valve inserted into the region of the PFO into the atrial septum, further contributing to its relative thickness. In total, the diameter measured 8 mm. A 25 mm device was thus chosen, as it was thought to adequately cover the length of the tunnel and had sufficient support on each side. The PFO was successfully closed using a 25 mm Gore Helex Septal Occluder (W.L. Gore and Associates, Flagstaff, Arizona) using previously described techniques. A post-procedural TEE confirmed appropriate positioning of the device, with both discs well deployed and no residual shunt, and the patient was discharged without complication.
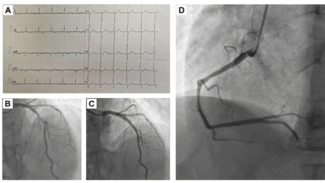
Three months following the initial procedure, it was noted that there was a fracture of Helex device’s locking loop when the patient was admitted for an unrelated procedure (Figure 2A). A subsequent TEE confirmed that the left atrial component of the device was poorly adherent to the interatrial septum, with some movement of the outermost portion of the left atrial disc during the cardiac cycle (Figure 2B). Associated with this was a large shunt. The device appeared stable, and we therefore initially elected to take a conservative approach with a bubble contrast study at follow up. The patient was advised not to dive until he was reviewed.
At follow up, imaging demonstrated persistent mobility of the outer portion of the left atrial disc, with a significant interatrial shunt. The patient was amenable to considering another procedure in order to return to diving. In addition, there was concern about device embolization with a continued conservative strategy, particularly if a wire frame fracture evolved because of the increased device mobility. The potential options were discussed with the patient (conservative versus catheter-based therapy versus surgery), and he expressed a strong desire for percutaneous intervention.
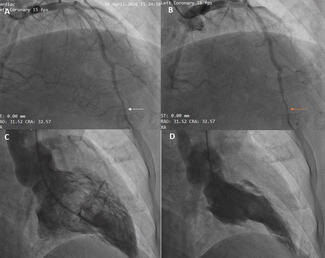
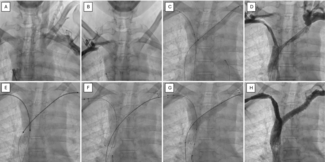
The procedure was performed under general anesthesia using fluoroscopic and TEE guidance. After placement of an 8 French (Fr) femoral sheath, a 5 Fr right Judkins coronary catheter (Cordis Corp., Miami Lakes, Florida) was used to probe the Helex device in an attempt to cannulate the left atrium through the residual leak. However, the terminal angle on the right Judkins was too acute, and we thus proceeded to use a Goodale-Lubin catheter (Medtronic, Inc., Minneapolis, Minnesota), which passed easily under the right atrial disc of the Helex device. A 0.035 inch exchange-length Terumo wire (Terumo Corp., Tokyo, Japan) was then used to gently probe the septum between the Helex device discs, and the left atrium was successfully cannulated. A 4 Fr Gensini catheter (Cordis), due to its good compliance and low profile, was exchanged over the Terumo wire and positioned into the left upper pulmonary vein. An exchange-length (260 cm) ordinary wire was then positioned in the left upper pulmonary vein, and an 8 Fr Amplatzer delivery system was then advanced into the left atrium. A 25 mm Cribriform Amplatzer device (AGA Medical Corp., Golden Valley, Minnesota) was delivered through an 8 Fr delivery system under TEE guidance. TEE imaging confirmed that the atrial septum was crossed between the Helex device discs and through the residual leak, rather than through a separate septal perforation. The Cribriform device was then deployed across the septum in such a way as to engage and hold on to the left atrial disc of the Helex device, including the locking stylet (Figure 3A). This was confirmed on TEE imaging. The right atrial disc was then deployed as distally from the left atrial disc as possible. The Cribriform right atrial disc was somewhat distorted by the adherent right atrial disc of the Helex device, such that the Cribriform device assumed a more teardrop shape facing inferiorly toward the inferior cava. This achieved stable position of both devices, and complete containment and stabilization of the left atrial disc of the Helex device (Figure 3B). There was no residual leak with Valsalva at the end of the procedure or at 6-week follow up.
Discussion. The safety of the Gore Helex septal occluder device has been evaluated in series enrolling 229 patients with PFOs.1,2 In these series, there were 2 cases of wire frame fracture, but no reports of locking-loop fracture. In addition to a recently published report,3 there has been only one other previous report of a locking-loop fracture with the Helex device.4 Though rare, locking-loop fracture is an significant complication because of the associated risk of residual shunting, thromboembolism and a concern about device stability and embolism, particularly should a wire-frame fracture also evolve because of the increased device mobility.
Anatomical differences in PFO anatomy are significant in predisposing patients to specific device-associated complications such as defects with poor margins which are likely to result in device embolization. In our case, the interatrial septum was relatively thick, with a long tunneled PFO. It is possible that this particular anatomical variation placed the locking loop under increased tension by transferring forces exerted by the contracting atrial myocardium directly to the locking loop, thus contributing to its fracture. The The Helex device was initially designed for closure of secundum atrial septal defects, though it is now licensed in Europe (but not in the United States) for PFO closure. In Europe, however, the manufacturers do not recommend its use for PFO closure in patients with a septal thickness > 8 mm in the area of occluder placement. The suitability of specific PFO anatomy in relation to the Helex device has not been formally evaluated. In cases similar to ours, with a relatively thick septum coupled to a long tunneled PFO, another device may be more suitable to avoid this rare, but potentially significant, complication. If faced with similar anatomy in the future, we would therefore advocate using a less compliant device that is more likely to distort the PFO anatomy to such a degree that the device’s predetermined shape is maintained. Such currently available devices include the Amplatzer PFO occluder (AGA Medical) and the Solysafe Septal Occluder (Swissimplant AG, Solothurn, Switzerland).3
When faced with locking-loop fractures, a number of potential management strategies are available. In the one previous case described, the fractured device was removed percutaneously 6 weeks after implantation because of right atrial disc mobility.4 There were no clinical sequelae. Additional options include a continued conservative strategy, with close observation of the device and patient, open surgical or transcatheter removal of the device, or, as in our case, insertion of another device percutaneously. Although cases of device embolization have been described in the literature, the absolute risk of locking-loop fracture evolving to wire-frame fracture and device embolization is uncertain.
In our case, an important reason for attempting another procedure was the significant residual interatrial shunt and the patient’s wish to resume diving. We pursued a percutaneous route of intervention, as this was the patient’s desire. We decided not to attempt to retrieve the Helex device, not because this was technically too difficult, but rather because of our concern regarding stability of the interlocking stylet on the left atrial side. Device retrieval through the atrial septum may well have dislodged and embolized the locking stylet.
Crossing of the residual leak was anticipated to be technically challenging. However, positioning of the probing catheter on the outer surface of the Helex right atrial disc, followed by gentle exploration with the Terumo wire, facilitated relatively straightforward access to the left atrium. Thus far, the outcome has been successful, with no short-term complications or residual shunting, and the patient plans to recommence diving. However, the longer-term safety is unknown.
For the second procedure, we chose a Cribriform Amplatzer Occluder (AGA Medical), a less compliant device as compared to the Helex. This recruited the left atrial disc well and stabilized it as well as the interlocking stylet very effectively, resulting in complete closure of the residual shunt. We felt this was the most important task to accomplish during the procedure, while the relative distortion of the right atrial disc was of lesser significance. Our experience with larger devices demonstrates that incomplete endothelialization may be a relatively frequent occurrence and does not necessarily increase device-related morbidity. The right atrial disc distortion was not out of keeping with that seen in patients requiring multiple overlapping devices.5 We felt that this was safe and that further device remodeling was likely to occur over time, as the device tends to assume it preprogrammed shape.
Conclusion. Though rare, locking-loop fracture of the Gore Helex Septal Occluder is a potentially significant complication that may lead to persistent inter-atrial shunting and device instability. Percutaneous insertion of a second closure device to stabilize a locking-loop fracture is both feasible and safe in the short term, and should be considered in cases such as ours, where there is a residual shunt and the patient wants to continue diving.