Thermal Imaging of Superficial Leg Circulation Improves Venous Diagnostic Efficiency and Completeness
VASCULAR DISEASE MANAGEMENT 2020;17(11):E208-E211
Abstract
Objective. Thermal imaging (TI) has been used in the medical field for over 50 years; however, it has shown limited clinical utility partly due to prohibitive costs, size, and often its use in inappropriate disease states. In this single center retrospective analysis, the impact of thermal imaging of legs was assessed by comparing complete duplex ultrasound (DUS) diagnostic results before and after the addition of TI to the routine clinical evaluation. Methods. One hundred and one consecutive patients were selected in 2017 before the introduction of routine TI prior to DUS and a second consecutive 101 patients were selected in 2019, following routine introduction of TI prior to DUS. All DUS were performed with the same machines, technicians and parameters used typical venous practice within a single institution. Charts were retrospectively reviewed and each leg considered separately. The DUS reports were reviewed independently by 2 experienced RVTs (sonographers) scoring each segment (GSV above the knee, GSV below the knee, anterior accessory great saphenous vein (AAGSV), posterior accessory great saphenous vein (PAGSV) and the small saphenous vein (SSV) as either positive for reflux (>0.5 seconds) or negative (<0.5 seconds). Results. The results before and after the introduction of TI were then compared: In 2017 there were 224 incompetent segments identified but in 2019 this had increased to 279, an increase of 24.6% which was statistically significant P<.015. Conclusions. In this study of 404 limbs undergoing venous investigation by DUS before and after the introduction of TI there was a statistically significant increase in the number of incompetent segments detected. Thus, the introduction of TI as a precursor to DUS resulted in more clinically relevant incompetent segments being detected.
Introduction
Thermal imaging has been available for many years but has failed to become established as a routine tool in any disease. Indeed in the past some investigators reported on thermal images and may have over claimed its diagnostic capability, most strikingly in breast cancer detection which resulted in the FDA issuing warning letters that thermal imaging should not be solely relied on in screening for breast cancer due to an excessive false negative rate, but may be used adjunctively along with conventional imaging modalities.1 This has resulted in the advancement of thermal imaging being slowed down even though there are CPT codes available for thermal imaging. Thermal imaging is based solely on the physical phenomenon that all objects above absolute zero emit heat energy, from the surface of the object, and the emitted energy is directly proportional to the surface temperature of the object. The thermal energy emitted can be focused and detected on a chip analogous to a digital camera and the temperatures displayed on a thermograph, typically color coded for differing temperatures.1 The emissivity of the surface of a material is its effectiveness in emitting energy as thermal radiation.3 Thermal radiation is a segment of the spectrum that includes both visible radiation (light) and infrared radiation, which is not visible to human eyes. It is precisely this infrared radiation that thermography exploits to yield clinically useful visible patterns.1,3
Perfect emissivity is 1.0 and human skin has an emissivity of 0.95-0.98, so for practical purposes in medical usage the skin emissivity is usually sufficiently close to unity to be considered a “black body” it has been determined that skin color does not influence emissivity in the mid-infrared spectrum.4-6 It is true that as anatomical structures are closer to the skin, the insulating effect of the subcutaneous tissue is less and more heat is conducted to the skin.4 The skin over these structures becomes relatively warm and is detectable with TI.
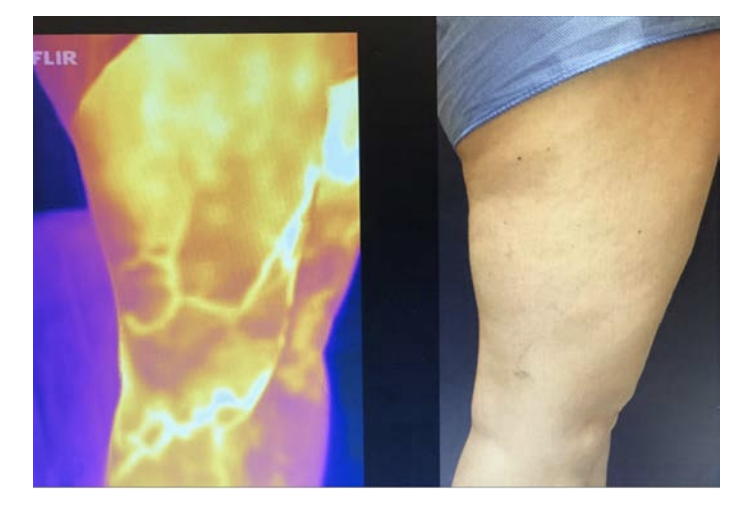
Thermal imaging as a possible tool for detection of superficial vein incompetence has been reported by several authors.7,8 TI was introduced as a routine in 2018 in the current study centre, a typical vein clinic and has become a helpful addition to the investigation of patients with signs and symptoms of chronic venous insufficiency. The perceived benefits are the creation of a superficial vein map and permanent record of the distribution of incompetent superficial veins and as a guide for the duplex technologist which focuses and speeds the complete duplex venous exam (Figure 3). As a highly visual medium it is easily understood and is a helpful tool in discussions with patients.
This retrospective chart-based study was devised to investigate if the introduction of TI had an impact on the detection rates of venous disease. During the time span of the study the venous technologists on staff remained constant and worked to the same protocol (IAC compliant), and practice referral patterns were unaltered. It was hypothesised that therefore any significant change in detection of venous disease on DUS might be attributable to the introduction of thermal imaging.
Method
This study was conducted in a high-volume single physician practice (Soffer Health Institute, PA)) in South Florida with a community-based referral pattern. The electronic charts of 2 groups of 100 consecutive patients attending the practice for investigation of signs and symptoms of chronic venous insufficiency were selected. One group in 2017 and the other after the introduction of TI in 2019. Patients were male or female, 21-70 years of age by year of birth and had complete DUS records. Patients with history of previous vein treatment surgery or ablation were excluded, previous sclerotherapy for cosmetic reasons was not an exclusion criterion. The DUS charts were reviewed independently by 2 ultrasound technologists and scored for the detection of incompetence with reflux >0.5 seconds in GSV above the knee, GSV below the knee, the anterior accessory GSV (AAGSV), the posterior accessory GSV (PAGSV) and the small saphenous vein (SSV). GSV incompetence detected in the thigh that did not propagate to the below knee segment or other superficial veins was considered negative. Each leg was considered separately thus there were 2 sets of 200 limbs to be compared.
Thermographic images were captured with the exposed leg with the patient standing, patients were not permitted to lie down prior to the image creation and walking was encouraged but not controlled. The images were captured by a FLIR One Pro™ (FLIR Systems, Inc. Wilsonville, Or) camera connected to an iPhone™ 8 or newer with iOS X or newer, and stored in a HIPPA compliant manner using RxPhoto™ software (AppWorx, Boston Ma). Routine ultrasound was conducted by qualified sonographers following ICAVL guidelines.
Statistical analysis was conducted by PH Nova Southeastern University Ft. Lauderdale, FL. We conducted a mixed-nested, logistic regression analysis. The independent variable was year (2017 vs 2019). Measurements for each leg were nested within the individual. Stata 16.1 was used for all data analysis. Statistical significance was considered found at P<0.05.
Results
Of the 202 patients selected, mean age was 49 in the 2017 group compared to 53 in the 2019 group; 81% of subjects were female in 2017 compared with 83% in 2019 and neither difference was statistically significant.
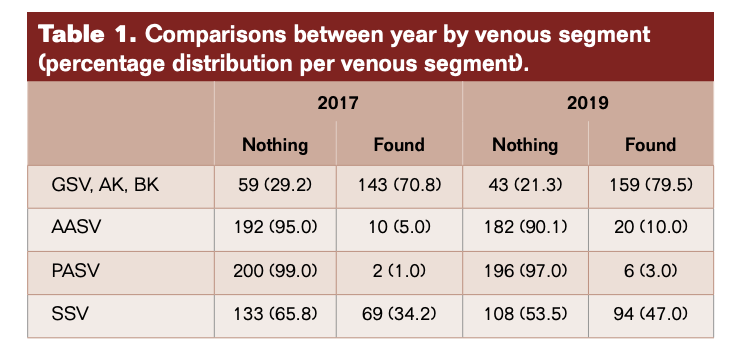
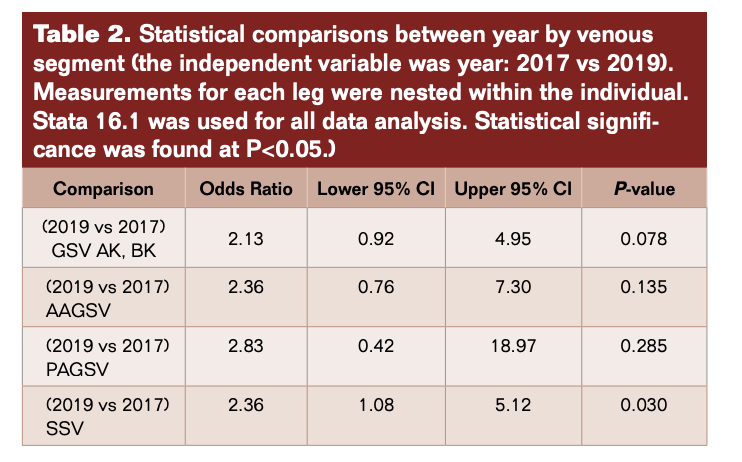
In 2017, reflux was found in 224 segments: 143 GSV, 10 AAGSV, 2 PAGSV, and 69 SSV compared with 279 segments and 159, 20, 6, 94, respectively in 2019 (Figure 1, Table 1). The overall difference in detection of incompetence was statistically significant P = 0.015 (95% CI 6.4-61). Considering the individual vein segments the difference in GSV was not significant (P=0.078) and while the frequency of detection of AAGSV doubled from 10 to 20 and PAGSV tripled from 2 to 6, the numbers are too small to be significant. There was a statistically significant increase in the detection of SSV incompetence of 36% (P=0.030) (95% CI 1.08-5.12) (Table 2).
Discussion
Thermal imaging was introduced in a single physician venous practice in 2018 and has since been adopted as an integral part of the patient work-up. The clinicians and ultrasound technologists find it to be a very user-friendly, time saving, and helpful resource for their venous patient examinations. They believe it improves the efficiency of DUS examination by significantly and consistently reducing the total exam time while improving anatomical venous mapping results. Because of its highly visual and recognizable imagery it has also been a useful tool for patient education, explanation, and ultimately compliance. A future prospective study is being considered to evaluate exactly how much time is saved with and without thermographic imaging and how the images positively affected the patient experience. Additionally, as venous leg ulcers continue to be a significant problem, studies to help wound centres identify patients and refer them to DUS is also being discussed.
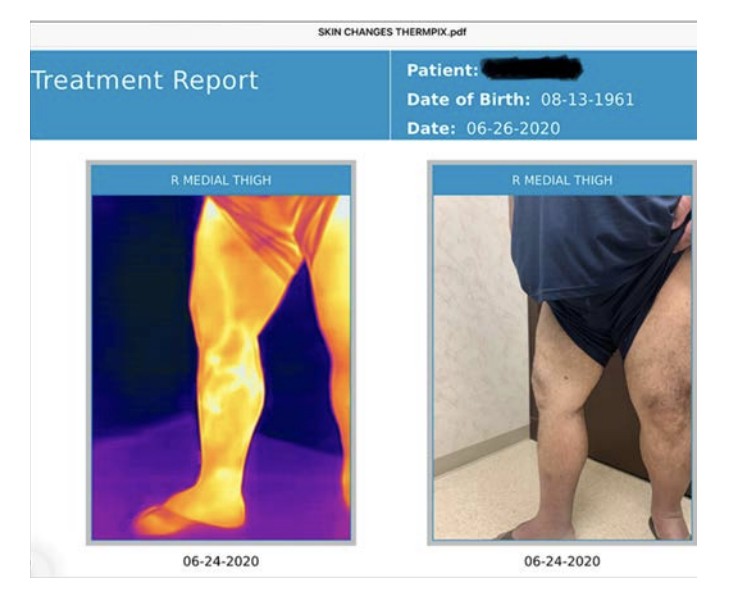
Over the decade 2010-2020, thermal imaging cameras have become more available, cheaper, smaller and more usable. In 2015 the authors started investigating the use of TI for superficial vein disease. It soon became obvious that varicose veins showed up very well on thermal imaging. Some authors have concluded that the increased heat over veins is caused by inflammation.4 We believe that we may be the first to describe that the increased heat pattern seen over the insufficient superficial veins is due instead to the core blood leaking toward the skin surface and not from inflammation. Venous blood in the deep veins has previously supplied the muscles and other structures deep to the deep fascia and is close to core body temperature (approximately 37°C). Conversely, venous blood in the normal superficial veins of the leg is blood that has been through the capillary network of the skin and subcutaneous tissue and so has equilibrated with skin temperature which is typically around 29-30°C.6 On thermal imaging normal (competent) superficial veins therefore do not show up as thermographically different from the skin and are therefore not detected. In superficial venous disease blood refluxes from the deep veins into the superficial veins and down the leg through incompetent valves, most commonly the great saphenous vein (GSV), the refluxing blood is substantially hotter than the skin and heat conducts to the surface and is detected as an area of elevated skin temperature that overlies the course of the incompetent superficial vein and is easily seen on TI (Figure 2 and 3 A&B). It has been suggested that the temperature differences are caused by inflammation of the vein, despite the fact that for simple varicose veins there are none of the markers of inflammation, redness and swelling of surrounding tissue, both of which are clearly seen in superficial thrombophebitis. Histological studies of simple varicose veins confirm this with the main finding being intimal hypertrophy and fibrosis with muscle thinning.9,10 In addition, if an individual is imaged while standing the vein pattern is very clear, 2-3 minutes after lying down the pattern is no longer present, if they stand again the vein pattern reappears. The authors believe this is clear evidence of the transient nature of the signal and its association with reflux. If it were caused by inflammation the pattern would be sustained for an extended period. The incompetent trunk vein is not usually seen as it lies deeper in its own fascial sheath. Adjacent structures to the incompetent truck vein are also at core body temperature and equally conduct heat to the surface so no excess heat is detected. Once the vein escapes the deep fascia and comes closer to the skin, the surface temperature is elevated and is typically around 33°C. This consistently creates a localised elevation in temperature along the incompetent vein of approximately 3°C when compared to nearly all adjacent anatomy, which is easily detected with modern TI cameras with sensitivity of 0.1°C.
It might be imagined that obese legs would not display vein patterns in the same way as slim individuals as the insulating effect of the thicker subcutaneous tissue was greater.4 In practice this does not seem to be the case, as the skin surface in the obese or edematous patient is likely to be cooler than that of a slim person, thus the relative temperature difference is maintained. While there has been no study of this reported in the literature the authors have not found thermal imaging of the obese leg to be any less useful (Figure 2 and 3).
To see if there was a change in diagnostic results following the introduction of thermal imaging, two consecutive patient series were compared. The first series was before the introduction for TI, and the second after TI was routinely and consistently used on all patients prior to DUS. Then the frequencies with which the major trunk vein segments were identified as incompetent were compared between the two-patient series. For all segments there were numerical increases after TI introduction and comparing all identified segments there was a 24.6% increase in trunk vein incompetence which was statistically significant. For the GSV while there was a modest increase in detection, that was not statistically significant. This was to be expected as TI is not usually capable of detecting the incompetent trunk vein in the thigh, but highly capable of detecting the GSV below the knee. The detection of incompetence from the AAGSV and the PAGSV were both increased by 2- and 3-fold, respectively but as the overall frequencies are low these failed to reach statistical significance in this relatively small series of patients. The trend toward increased detection of incompetent AAGSV and PAGSV was not a surprise to the investigators as they tend to run closer to the surface than the truncal GSV. TI can detect lateral and upper posterior thigh vein incompetence which guides the sonographer to look in areas that might otherwise be less obvious and is often missed on routine exam. Additionally, it has served as a very beneficial tool for patient education, allowing visual discussion of venous status that is much more visually comprehensible to the patient than traditional DUS data (Figure 3).
This study has obvious limitations as a retrospective, electronic chart-based review. The investigators agree that there were no changes to the types of protocols, personnel and referral patterns which might account for the changes in detection rates and therefore the most likely material change was the introduction of thermal imaging. It should be noted that it does not imply direct causal relationship as a result of the images themselves.
In summary, Thermal Imaging introduction to a vascular practice has resulted in a 24.6% increase in clinically relevant incompetent trunk vein detection resulting in a commensurate increase in clinically appropriate procedures which in turn could be expected to improve the clinical outcome of that patient population. It is the investigators conclusion that adding Thermal Imaging to a clinical practice appears to increase the completeness and efficiency of the duplex ultrasound examination, but future prospective multi-center trials are required to confirm these findings.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Marcus Caine is a paid consultant of USA Therm LLC.David DI Wright and Ariel Soffer are founding directors of USA Therm LLC. All other authors have nothing to disclose.
Address for Correspondence: Ariel D. Soffer, Email: as@sofferhealth.com
REFERENCES
1. Lozano A, Hayes JC, Compton LM, Azarnoosh J, Hassanipour F. Determining the thermal characteristics of breast cancer based on high-resolution infrared imaging, 3D breast scans, and magnetic resonance imaging. Sci Rep. 2020;10(1):10105.
2. Singh D, Singh AK. Role of image thermography in early breast cancer detection- Past, present and future. Comput Methods Programs Biomed. 2020;183:105074.
3. Cervantes AM, Machuca ESK, Guevara E, González FJ, Flores JJ. Evaluation of Breast Cancer by Infrared Thermography. Published online 2020:13.
4. Hejazi S, Wobschall DC, M.d RAS, Anbar M. Scope and limitations of thermal imaging using multiwavelength infrared detection. Opt Eng. 1992;31(11):2383-2393.
5. Steketee J. Spectral emissivity of skin and pericardium. Phys Med Biol. 1973;18(5):686–694.
6. Sanchez-Marin FJ, Calixto-Carrera S, Villaseñor-Mora C. Novel approach to assess the emissivity of the human skin. J Biomed Opt. 2009;14(2):024006.
7. Cholewka A, Stanek A, Klimas A, Sieroń A, Drzazga Z. Thermal imaging application in chronic venous disease. J Therm Anal Calorim. 2014;115(2):1609-1618.
8. Alvarado Díaz W, Roman Gonzalez A, Meneses Claudio B. Detection of suspicions of Varicose veins in the legs using thermal imaging. Repos Inst - UCH. Published online 2019.
9. Milroy CM, Scott DJA, Beard JD, Horrocks M, Bradfield JWB. Histological appearances of the long saphenous vein. J Pathol. 1989;159(4):311-316.
10. Wali M, Dewan M, Eid R. Histopathological changes in the wall of varicose veins. Int Angiol J Int Union Angiol. 2003;22:188-193.










