Application of 3 Different Types of Pedicled Adipofascial Flaps in the Repair of Deep Dead Space Wounds
Abstract
Introduction. Dead space is an important risk factor for poor wound healing; therefore, it is important to effectively fill deep dead space through individualized tissue flap design during the repair of complex wounds. Adipofascial flaps have yielded good results in the repair of deep dead space wounds. Objective. The authors evaluated the efficacy of 3 kinds of adipofascial flaps to repair deep dead space wounds. Methods. From January 2019 to January 2022, 15 patients with complicated wounds accompanied by deep dead space underwent repair via 1 of 3 kinds of adipofascial flaps, and the clinical efficacy was observed. Results. All 15 transplanted adipofascial flaps exhibited complete survival, and within a mean follow-up of 14.7 months, both the donor and recipient sites had successfully healed. Conclusion. The traditional pedicled adipofascial flap was used to repair single deep dead space wounds, and pedicled perforator adipofascial extension flaps or layered fasciocutaneous flaps were used on compound tissue defect wounds, thus providing a relatively simple, safe, and effective method to repair a small area of tissue defect with deep dead space wounds.
Introduction
Dead space is an important risk factor for poor wound healing; therefore, an important step in the repair of complex wounds is to effectively fill deep dead space through individualized tissue flap design.1,2 Like the muscle flap, both pedicled and free adipofascial flaps have achieved reliable results for the repair of deep dead space wounds.2-6
In the authors’ institution, 3 different types of pedicled adipofascial flaps are used to repair deep dead space wounds: traditional pedicled adipofascial flap, pedicled adipofascial extension flap, and pedicled layered fasciocutaneous flap. The traditional pedicled adipofascial flap is used to repair individual deep dead space wounds; however, if the repair of a complex wound does not require functional reconstruction (such as a small bone or deep tissue defect cavity with soft tissue defect), the pedicled adipofascial extension flap or pedicled layered fasciocutaneous flap can be used. The latter 2 kinds of adipofascial flaps are used to simultaneously repair skin and soft tissue defects with deep dead space wounds in a single step.
Here, the design and operative technique of these 3 flaps are presented, along with 15 cases describing the outcomes and effects of this approach.
Materials and Methods
Patients and treatment
The authors conducted a retrospective analysis of 15 patients who a) presented with either deep dead space wounds or complex defects with deep dead space, and b) underwent adipofascial flap repair; 5 patients were female and 10 patients were male (mean age, 39.2 years; range, 14-75 years). All patients were informed about the procedure and gave their written informed consent. This retrospective study was approved by the ethics committee of the North District of the First Affiliated Hospital of Anhui Medical University (PJ-YX2022-006). The size of skin and soft tissue defects ranged from 4 cm × 2 cm to 10 cm × 4 cm after debridement, while the size of deep dead space wounds ranged from 1.5 cm × 0.5 cm × 1.5 cm to 6 cm × 2 cm × 2 cm after debridement and NPWT. Seven patients were treated with traditional pedicled adipofascial flaps; in this group, causes of injury included traffic accidents (n = 4; sinus tract formation due to infection at the surgical incision or rejection of the ligament braid), indirect inguinal hernia (n = 1; sinus tract formation due to rejection of the patch), pressure injuries (n = 1), and a herniated lumbar intervertebral disc (n = 1; sinus tract formation due to infection at the surgical incision). Three patients were treated with pedicled adipofascial extension flaps; in this group, causes of injury included a fall (n = 1; sinus tract formation due to infection at the surgical incision) and pressure injuries (n = 2). Five patients were treated with pedicled layered fasciocutaneous flaps; in this group, causes of injury included traffic accidents (n = 2; sinus tract formation due to tissue necrosis after trauma), fall injuries (n = 1; tissue necrosis after trauma), pressure injuries (n = 1), and thoracic abscess (n = 1; tissue necrosis after infection).
Cultures showed bacterial growth in 5 patients who received traditional pedicled adipofascial flaps, 3 who received pedicled adipofascial extension flaps, and 2 who received pedicled layered fasciocutaneous flaps. Details of all 15 patients—including sex, age, cause of injury, defect site, soft tissue defect size, deep dead space defect size, skin paddle flap size, adipofascial flap size, bacterial culture result, donor site closure, and follow-up data (length of stay, number of operations)—are shown in the eTable.
Radical debridement of all necrotic and devitalized tissues was performed; if the wound required serial debridement, NPWT was applied for 3 to 5 days between debridement procedures until the wound was clear and ready for coverage. Empirical broad-spectrum antimicrobial therapy was initiated immediately and later modified based on tissue culture results, if necessary, and all patients underwent computed tomographic angiography to confirm that the blood vessels supplying the tissue flap to be selected were unobstructed.
Flap design and operative procedures
Traditional pedicled adipofascial flap
First, the skin along the midline overlying the adipofascial flap was incised, and the skin (along with a thin layer of fat approximately 1 mm thick) was sharply dissected and saved for later use. Second, starting at the distal end, the adipofascial flap was dissected from the subfascial level using a surgical blade and scissor. The adipofascial flap was rotated or turned over 180 degrees and packed into the deep dead space; the wound and donor area could then be directly sutured.
Pedicled adipofascial extension flap
First, Doppler ultrasound was used to detect perforators of the target artery. Second, the skin along the midline overlying the adipofascial extension was incised, and the skin (together with a thin layer of fat approximately 1 mm thick) was sharply dissected without saving. Third, starting at the distal end of the adipofascial extension, the flap was dissected from the subfascial level using a surgical blade and scissor. The adipofascial flap was inserted or gently curled in a vertical fashion to fill the deep dead space, and the main part of the fasciocutaneous skin flap was shifted to the lateral edge of the wounds to cover and seal all exposed wounds. The donor area could be closed directly, or a large split-thickness skin graft was transplanted to repair the donor area wounds, if necessary.
Pedicled layered fasciocutaneous flap
First, Doppler ultrasound was used to detect perforators of the target artery. Second, the fasciocutaneous flap with the target pedicled perforator artery was excised. Third, starting at the distal end of the primary flap, the flap was dissected from the fat level (about 2 mm under the superficial fascia) using a surgical blade and scissor; the sharp separation was about 3 to 4 cm lengthwise or transverse. The primary flap was then divided into a skin paddle and an adipofascial flap. The skin paddle contained the epidermis, dermis, superficial fascia, and shallow fat layer, which was approximately 2 mm below the superficial fascia. The adipofascial flap included the deep fat layer, which was approximately 2 mm below the superficial fascia, and deep fascia.
The adipofascial flap was inserted or gently curled in a vertical fashion to fill the deep dead space. Subsequently, the skin paddle was shifted to the lateral edge of the wound to cover all exposed wounds. A large split-thickness skin graft was transplanted to repair the donor area wounds.
Results
All 15 transplanted adipofascial flaps demonstrated complete survival. The distal end of the skin paddle in case 12 had congestion of 1 cm × 1 cm in size; superficial skin ulceration occurred, which healed after dressing change. The skin grafts in the donor area of cases 14 and 15 showed slight marginal epidermal necrosis changes, and the wounds healed after 2 weeks of dressing change. The mean follow-up time was 14.7 months. No relapses of infection were observed, and all patients demonstrated good long-term coverage, flap pliability, and contour at the flap site.
Case 1
A 72-year-old male underwent surgery for a herniated lumbar intervertebral disc 5 months before admission. After being discharged from the hospital, he remained bedridden for approximately 2 weeks, resulting in the formation of pressure injuries on the sacrococcygeal skin. Although the dressings on the pressure injuries were regularly changed in the clinic, the wounds did not significantly improve. Approximately 4.5 months later, fever of 38.6°C (101.5°F) developed; he received antibiotic treatment at another hospital but still exhibited intermittent fever with a maximum body temperature of 39°C (102.2°F).
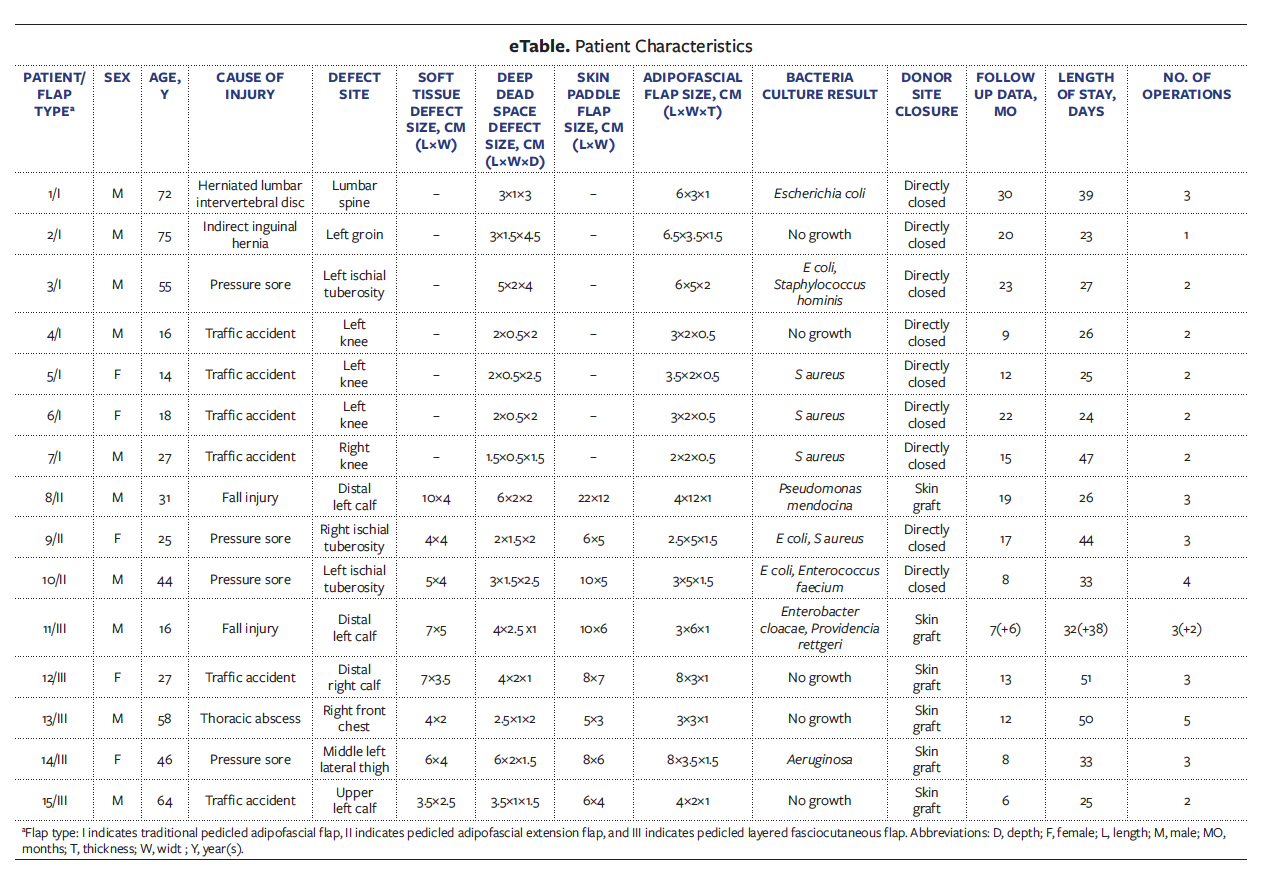
The patient presented to the authors’ hospital with a pressure injury measuring 9 cm × 5 cm in size, visible on the sacrococcygeal skin; the skin behind the lumbar spine showed redness and swelling (Figure 1A, taken 1 day after admission). Antibiotic treatment was administered. The wounds were disinfected with iodophor, covered with iodoform gauze, and externally covered with gauze for protection. The wounds were disinfected, and the dressings were changed once daily until the operation was performed.
Debridement surgery was performed on the pressure injuries located in the sacral coccyx, and NPWT was used on Day 4 of hospitalization; however, the patient’s lumbar spine surgical scar remained red and swollen. On Day 7, a puncture was made, and purulence accumulation was evident (Figure 1B); therefore, exploration and debridement were conducted on the wounds. The dead space was found to be about 5 cm deep, and internal fixation was detectable (Figure 1C, taken on Day 9); NPWT was used in the deep dead space.
After debridement and antibiotic treatment, bacterial cultures of the wounds on the back of the lumbar vertebrae showed no growth. Fresh granulation tissue had formed, and the deep dead space reduced to approximately 3 cm × 1 cm × 3 cm (Figure 1D, Day 14). A pedicled adipofascial flap measuring approximately 6 cm × 3 cm × 1 cm was designed that filled the dead space after being turned over 180 degrees (Figure 1E, Day 14). The skin over the wound was directly pulled (advanced), and the skin and donor area were directly sutured. Drainage tubes formed by manual processing of an ordinary disposable infusion set (4.5 mm, type B1-1) were then placed in the dead space and donor area (Figure 1F, Day 14).
When the drainage fluid volume was less than 2 mL for 3 consecutive days and became serous, the drainage tube could be removed. The left drainage tube was removed 4 days after the procedure, and the right drainage tube was removed on the eighth postoperative day. During this period, the wounds were routinely disinfected with iodophor and dressings were changed.
On evaluation 30 months after the procedure, there was no recurrence in the posterior lumbar area or sacrococcygeal skin, and the scars formed were acceptable to the patient (Figure 1G).
Case 8
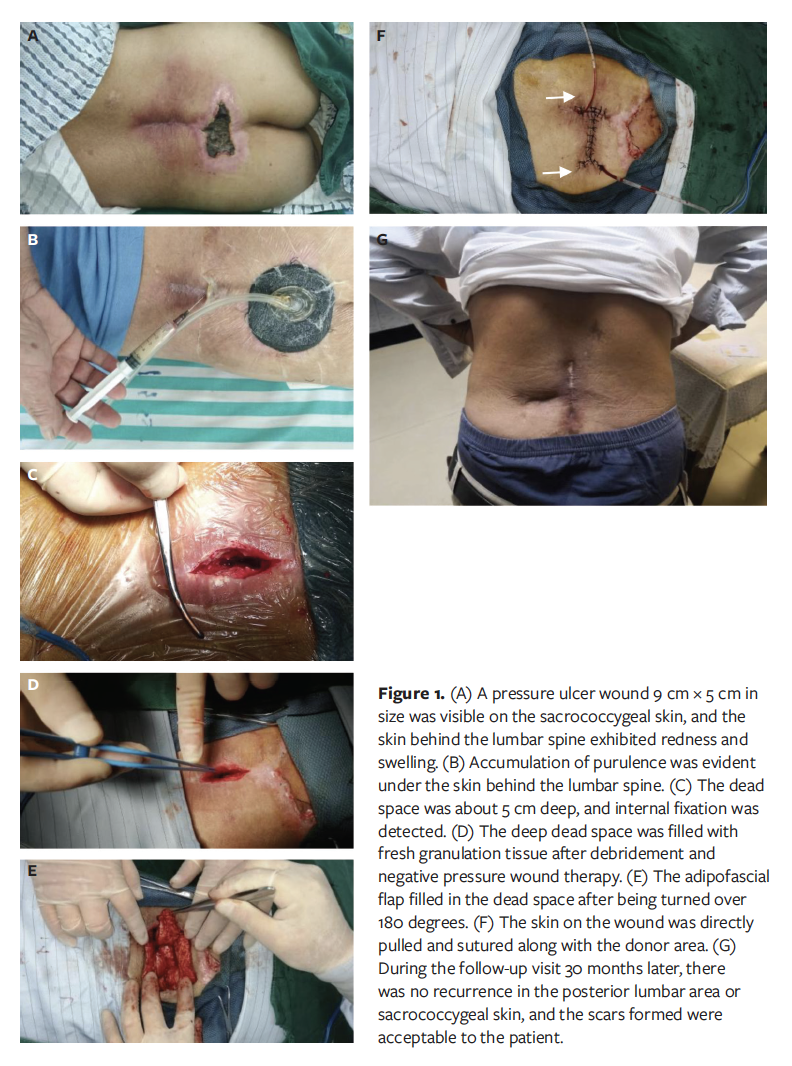
A 31-year-old male underwent open reduction and internal fixation of left tibial and fibular fractures sustained in a fall. One week after the operation, skin necrosis and extensor tendon exposure gradually appeared at the incisional site, and he was admitted to the authors’ hospital. During the first surgical debridement, the fatty and connective tissue between the extensor tendons were found to have degenerated, and ischemic necrosis had developed (Figure 2A). After radical debridement, the implants were exposed with a deep dead space (Figure 2B), and NPWT was used on the wounds.
After debridement and antibiotic treatment, bacterial culture of the wounds showed no growth. Fresh granulation tissue had formed at the wound bed. The soft-tissue defect on the lower extremity measured 10 cm × 4 cm, while the deep dead space was 6 cm × 2 cm × 2 cm. An adipofascial extension flap with pedicled posterior tibial artery perforator was harvested (Figure 2C), the main part of the fasciocutaneous flap was used to cover the exposed wound, and the distal adipofascial flap was put into the dead space to obliterate the deep dead space (Figure 2D). A large split-thickness skin graft was transplanted to repair the donor area wounds. At the 19-month follow-up, there was no sign of recurrence of necrosis or infection at the wound repair site, and no local venous reflux disturbances were observed in the leg (Figure 2E).
Case 11
A 16-year-old male sustained left tibial and fibular fractures with skin and soft tissue necrosis following a fall. The patient underwent debridement surgery in another hospital and subsequently underwent external fixation reduction for the tibial and fibula fractures. NPWT was used on the patient’s wound sites.
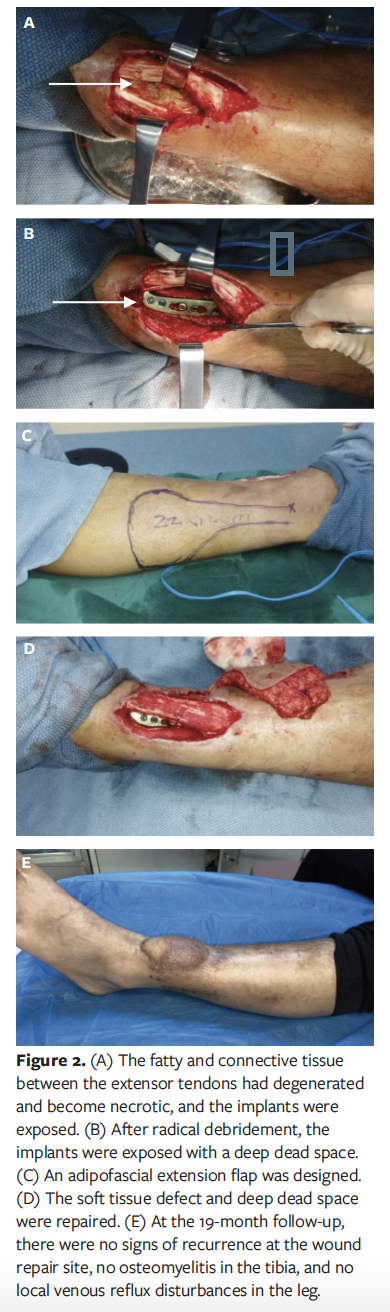
When the patient was admitted to the authors’ hospital, there were skin and soft tissue defects with necrosis of peroneus longus and peroneus brevis tendons on the lower part of the lateral and posterior fibula, and the wound bed was covered with fresh granulation tissue (Figure 3A). After debridement and antibiotic treatment, bacterial culture of the wounds showed no growth. Fresh granulation tissue had formed at the wound bed and was appropriated for repair. The fibula and tibia bones were exposed with a small amount of bone defect seen at the tibial fracture site. The soft-tissue defect on the lower calf measured 7 cm × 5 cm, and the deep dead space was 4 cm × 2.5 cm × 1 cm (Figure 3B). The tibial bone defect was filled with 3 bone cements prepared with vancomycin (Figure 3C). A layered faciocutaneous flap with pedicled peroneal artery perforator from the posterolateral side of the ipsilateral fibula was harvested, the skin paddle was used to cover the exposed wounds, and the distal adipofascial flap was used to obliterate the deep dead space. A large split-thickness skin graft was transplanted to repair the donor area wounds (Figure 3D). After all of the wounds had healed, the patient was discharged.
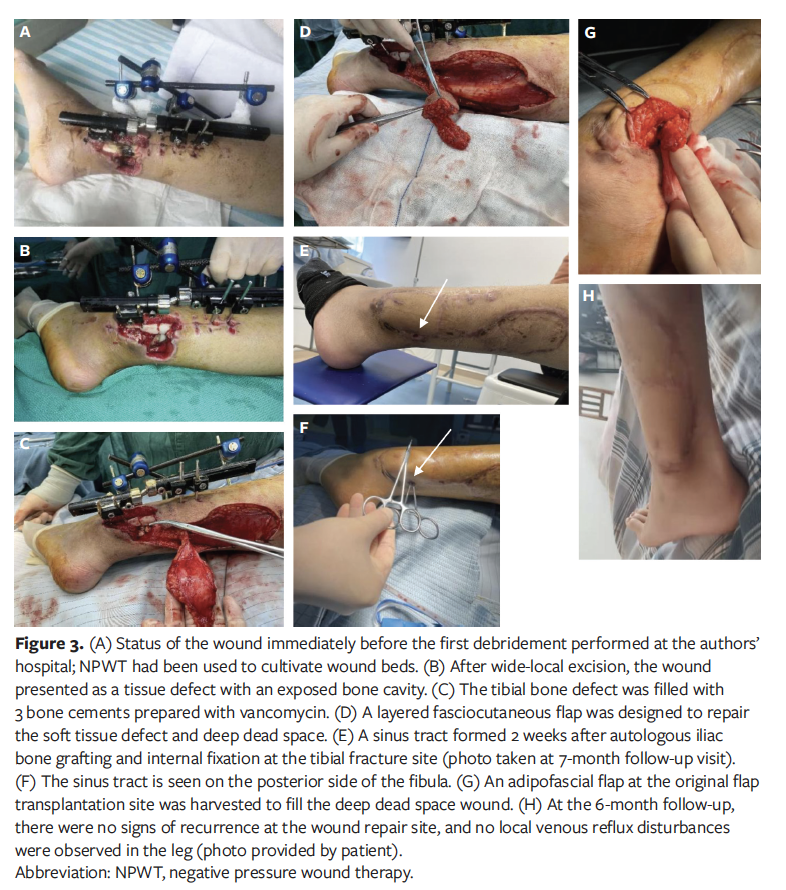
Seven months after discharge, the patient was admitted to a different hospital for removal of the external fixator of the left tibia and fibula and to undergo autologous iliac bone grafting and internal fixation at the tibial fracture site. Two weeks after surgery, an incision on the posterior side of the fibula had not healed, and a sinus tract had formed (Figure 3E). The patient was then readmitted to the authors’ hospital for treatment. The sinus tract on the posterior side of the fibula measured approximately 4 cm in depth (Figure 3F). After antibiotic therapy and NPWT, a 5 cm × 1.5 cm adipofascial flap at the original flap transplantation site was harvested to fill the deep dead space wounds, and the donor flap and the skin at the wounds was directly closed by suture (Figure 3G). On evaluation 6 months later, there were no signs of recurrence at the wound repair site, and there was no local venous reflux disturbance in the leg (Figure 3H).
Discussion
Complicated wounds often manifest as compound tissue defects, referring to the presence of 2 or more kinds of tissue defects occurring simultaneously.1,7 Deep dead space is an important risk factor for poor wound healing.1,2 Eliminating deep dead space is important to repairing compound tissue defects but has always posed a problem to clinicians1,7; therefore, in the repair of complicated wounds, it is crucial to effectively fill deep dead space wounds through individualized tissue flap design.1,2
An improved understanding of the perforator and vascular anatomy, coupled with advances in microsurgery, have resulted in the recent development of the chimeric flap technique. The chimeric flap has separate components, with separate vascular supplies attached to a common pedicle.8 Chimeric flaps can be used to reconstruct complicated or extensive defects while maintaining efficient tissue usage and low donor morbidity.1,7,9,10 These chimeric tissue flaps, either with1,7,9 or without10 muscle tissue, can better repair complex wounds with deep dead space. During the application of other surgical methods such as the muscle flap,11-13 both pedicled and free adipofascial flaps have achieved good results in the repair of deep dead space wounds.2-6
Although the previously described free adipofascial flaps can be used to repair complex wounds,2,5 this kind of repair surgery usually requires the surgeon to have more proficient micromanipulation techniques.2,5 When the repair of complex wounds does not require functional reconstruction, layered fasciocutaneous flaps or adipofascial extension flaps with pedicled perforator can be used to repair these types of wounds. Unlike the free adipofascial flap operation, the pedicled blood vessel does not need to be freed and anastomosed; thus, the procedure is relatively simple and safe.14
In this retrospective analysis, 3 types of pedicled adipofascial flaps were used to repair deep dead space wounds: traditional pedicled adipofascial flaps, pedicled adipofascial extension flaps, and pedicled layered fasciocutaneous flaps. Traditional pedicled adipofascial flaps were used to repair single deep dead space wounds; this type of surgery has been widely used in clinical practice. Recently, pedicled adipofascial extension flaps have gradually been used in the repair of complex wounds.4,6 This kind of adipofascial flap was used to simultaneously repair skin and soft tissue defects and deep dead space wounds in one step.4,6
The pedicled layered fasciocutaneous flap can be seen as a modified version of the pedicled adipofascial extension flap, which retains the full-thickness skin tissue at the distal end or lateral side of the fasciocutaneous flap. To date, there is no precise definition of a pedicled layered fasciocutaneous flap, nor has a consensus been reached.14 Here, layered fasciocutaneous flaps were used to repair complex wounds with deep dead space in the front chest, buttocks, and lower limbs. In these operations, the superficial skin tissue and deep adipofascial tissue were nourished by different branches originated from the same blood vessel, and the primary fasciocutaneous flap could then be divided into a superficial skin paddle and a deep adipofascial flap. Consequently, the skin defect and deep dead space could be simultaneously repaired in one step.
When the perforating vessels pass through the muscle or intermuscular space, the divided grade II perforator vessels independently nourish the skin and subcutaneous adipofascial layer.15 The skin flap is rich in subdermal vascular networks consisting of ascending and horizontal branches of the perforator artery. Additionally, the blood supply of the deep fascia and deep fat is independent of the blood supply to the superficial fascia; thus, after carefully and acutely separating the skin paddle from the fat level (found approximately 2 mm under the superficial fascia), the adipofascial flap retains a blood supply by preserving the subfascial plexus.16 The primary flap is divided into a double-layered flap.
The authors of the present study believe that pedicled adipofascial extension flaps are more suitable for complex wounds with deep dead space around the skin defect wounds (such as those in case 8), while pedicled layered fasciocutaneous flaps are more suitable for complex wounds with deep dead space at the center of the skin defect wounds (as seen in case 11). Because the donor area requires relatively rich adipose tissue, particular attention should be paid to designing a layered fasciocutaneous flap so that the superficial flap retains 2 mm of superficial fat; in addition, the deep adipofascial flap should not be too thin (preferably >0.5 cm).
Limitations
This case series has several limitations, one of which is the small number of patients treated. Second, whether the perforator vessel is carried or not will affect the length of the adipofascial flap. Third, the skin paddle of the layered fasciocutaneous flap is more prone to necrosis than that of the adipofascial flap, so effective intervention is needed to promote the survival of the skin paddle. Along those lines, the authors are exploring the use of platelet-rich plasma combined with layered fasciocutaneous flap to repair complex wounds.
Conclusions
Three types of pedicled adipofascial flaps can be used to repair deep dead space wounds. In this retrospective analysis, the traditional pedicled adipofascial flap was used to repair a single deep dead space wound, while pedicled adipofascial extension flaps or pedicled layered fasciocutaneous flaps were used to repair compound tissue defect wounds, providing a relatively simple, safe, and effective method to repair small tissue defects with deep dead space wounds. Although the pedicled adipofascial flap can provide an optimized method for wound repair, it should be noted that the vascular anastomosis technique may also need to be used. Nevertheless, larger randomized, controlled clinical trials and long-term follow-up are required to support the recommendation of the pedicled layered fasciocutaneous flap.
Acknowledgments
Authors: Zhengguo Xia, MD1,2; Pengtao Li, MSM3; Yin Wang, MD1,2; Wenting Wang, MSM1,2; Chao Hu, MSM1,2; Xinwang Cao, MD4; and Linsen Fang, MSM1,2
Acknowledgments: Z.X. and P.L. contributed equally to this work. This work was supported by the Scientific Research Fund Project of Anhui Medical University (2019xkj156), Peak Discipline Construction Project in Clinical Medicine of Anhui Medical University (9301001810).
Affiliations: 1Department of Wound Repair & Plastic and Aesthetic Surgery, the First Affiliated Hospital of Anhui Medical University, Anhui, China; 2Anhui Public Health Clinical Center, Anhui, China; 3Department of Plastic and Aesthetic Surgery, the First Affiliated Hospital of Anhui Medical University, Anhui, China; 4Department of Cell Biology, School of Life Sciences, Anhui Medical University, Anhui, China.
Disclosure: The authors disclose no financial or other conflicts of interest.
Correspondence: Linsen Fang, MSM, Department of Wound Repair & Plastic and Aesthetic Surgery, the First Affiliated Hospital of Anhui Medical University, No.100 Huaihai Rd., Xinzhan District, Hefei City, Anhui Province 230022, China; shaoshangke@126.com
References
1. Ren GH, Xiang DY, Wu XH, Chen YB, Li R. A neglected problem in the utilization of free anterolateral thigh flap toward reconstructing complicated wounds of extremities: the obliteration of deep dead space. J Orthop Surg Res. 2020;15(1):483. doi:10.1186/s13018-020-01914-0
2. Del Piñal F, Urrutia E, Klich M. Severe crush injury to the forearm and hand: The role of microsurgery. Clin Plast Surg. 2017;44(2):233-255. doi:10.1016/j.cps.2016.11.002
3. Schmidt K, Jakubietz MG, Gilbert F, Fenwick A, Meffert RH, Jakubietz RG. Muscle cuff in distal pedicled adipofascial sural artery flaps: a retrospective case control study. Plast Reconstr Surg Glob Open. 2021;9(3):e3464. doi:10.1097/GOX.0000000000003464
4. Zheng L, Zheng J, Dong ZG. Reverse sural flap with an adipofascial extension for reconstruction of soft tissue defects with dead spaces in the heel and ankle. Eur J Trauma Emerg Surg. 2016;42(4):503-511. doi:10.1007/s00068-015-0569-x
5. Mashrah MA, Mai L, Wan Q, et al. Posterior tibial artery flap with an adipofascial extension: clinical application in head and neck reconstruction with detailed insight into septocutaneous perforators and donor-site morbidity. Plast Reconstr Surg. 2020;145(1):142e-152e. doi:10.1097/PRS.0000000000006396
6. Peng P, Dong ZG, Liu L, Wei JW, Luo Z, Cao S. An effective technique for managing the calcaneus osteomyelitis combined with soft-tissue defect [published online ahead of print, 2021 May 3]. Int J Low Extrem Wounds. 2021;15347346211016696. doi:10.1177/15347346211016696
7. Sung IH, Jang DW, Kim SW, Kim YH, Kim SW. Reconstruction of diabetic lower leg and foot soft tissue defects using thoracodorsal artery perforator chimeric flaps. Microsurgery. 2018;38(6):674-681. doi:10.1002/micr.30314
8. Hallock GG. The complete nomenclature for combined perforator flaps. Plast Reconstr Surg. 2011;127(4):1720-1729. doi:10.1097/PRS.0b013e31820a662b
9. Qing LM, Wu PF, Yu F, Zhou ZB, Tang JY. Use of a sequential chimeric perforator flap for one-stage reconstruction of complex soft tissue defects of the extremities. Microsurgery. 2020;40(2):167-174. doi:10.1002/micr.30450
10. Guo EQ, Xie QP, Zhu ZG, Jin PH, Jin PH, Wang L. Laparoscopy-assisted chimeric peritoneal-deep inferior epigastric perforator flap for reconstruction of hand and foot. Wounds. 2018;30(2):36-40.
11. Cho EH, Shammas RL, Carney MJ, et al. Muscle versus fasciocutaneous free flaps in lower extremity traumatic reconstruction: a multicenter outcomes analysis. Plast Reconstr Surg. 2018;141(1):191-199. doi:10.1097/PRS.0000000000003927
12. Buono P, Castus P, Dubois-Ferrière V, et al. Muscular versus non-muscular free flaps for soft tissue coverage of chronic tibial osteomyelitis. World J Plast Surg. 2018;7(3):294-300. doi:10.29252/wjps.7.3.294
13. Garcia Del Pozo E, Collazos J, Carton JA, Camporro D, Asensi V. Factors predictive of relapse in adult bacterial osteomyelitis of long bones. BMC Infect Dis. 2018;18(1):635. doi:10.1186/s12879-018-3550-6
14. Xia ZG, Wang CH, Arnold AA, et al. Efficacy of treating chronic tibial osteomyelitis with bone defect using a pedicled perforator-layered flap and fasciocutaneous flap of the posterior tibial artery: a case report. Wounds. 2020;32(11):E50-E54.
15. Deng CL, Wu BH, Wei ZR, Li H, Zhang TH, Wang DL. Interperforator flow pattern and clinical application of distal extended peroneal artery perforator flaps. Ann Plast Surg. 2018;80(5):546-552. doi:10.1097/SAP.0000000000001290
16. Sun CS, Su B, Li WJ et al. Anatomic basis and clinical application of perforator flap thinning of posterior tibial artery. Article in Chinese. Chin J Microsurg. 2019;42(1):42-45. doi:10.3760/cma.j.issn.1001-2036.2019.01.011

















