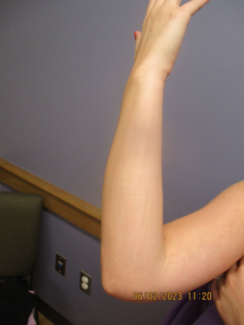
T he recent development of a foam vehicle for application of topical corticosteroids has proven beneficial for the treatment of scalp eruptions such as psoriasis and seborrheic dermatitis. Studies have demonstrated enhanced penetration and a high level of cosmetic acceptability as a result of rapid disappearance and penetration of the foam into skin without a residue. In addition to FDA-approved usage on scalp, further experience has shown that the foam vehicle may be used to treat other cutaneous body sites with successful results demonstrated for a wide variety of steroid responsive dermatoses. Here’s a look at the research supporting use of the foam vehicle and various clinical applications through illustrative case studies. Development And Evolution Of The Foam Vehicle Presently, two topical corticosteroids are available in the United States in a specially formulated foam vehicle. Betamethasone valerate 0.12% foam (BMV foam) has been available for use since FDA approval in 1999 and clobetasol propionate 0.05 % foam (CP foam) since approval in 2000. Other compounds also demonstrate stability in the foam vehicle and are currently under development. The foam formulation has been described in the literature as a novel vehicle with enhanced delivery.1,2 Enhanced delivery refers to rapid product dispersal upon application and enhanced percutaneous absorption in human skin models compared to other vehicle types. Prior to contact with skin, the foam remains stable. Upon skin contact, natural body heat causes rapid volatilization of inert contents with deposition of active drug onto the skin surface. In order to facilitate efficient use, it’s recommended that the patient place a small amount of the foam into the cap prior to application (see photo on page 76). As the foam contains <4. 5% solid ingredients, there’s little-to-no detectable residue on skin.1-3 “Quick break” dispersal also allows for easy spreadability, which is especially important when applying the foam to an extensive body surface area or intertriginous site. In studies using human skin, enhanced percutaneous penetration has been demonstrated in vitro with both BMV foam and CP foam. Within the first 24 hours, BMV foam exhibited six-fold greater skin penetration than betamethasone valerate 0.1% lotion (BMV lotion).1 In a comparison of CP foam and clobetasol propionate 0.05% solution (CP solution), CP foam demonstrated statistically significant greater peak and total absorption after both 12 hours and 48 hours.2 Vasoconstrictor assay studies revealed BMV foam as medium potency and CP foam as ultra-high potency. Clinical Efficacy Double-blind, placebo-controlled clinical studies have confirmed the efficacy of both corticosteroid foam formulations for moderate to severe scalp psoriasis in adults. These studies included active drug comparisons using other vehicles. Here’s a look at results from these studies. • Based on investigator global assessment after 28 days of active drug use, patients were judged to be “clear or almost clear” in 72% of patients treated with BMV foam (n = 57) and 47% treated with BMV lotion (n = 58).1 • Similar evaluation comparing 14 days of active treatment with CP foam (n = 62) or CP solution (n = 63) rated 74% of patients as “completely clear or almost clear” in the foam-treated group and 63% in the solution-treated group.2 Signs and symptoms of psoriasis demonstrated greater response after both 1 and 2 weeks of active therapy and at follow-up 2 weeks post-therapy in patients treated with CP foam as compared to CP solution or placebo. The adverse reaction profile of both foam compounds has proven to be favorable in multiple studies, and comparable to placebo and other tested comparator vehicles. • The efficacy of BMV foam for non-scalp psoriasis (n = 37) was confirmed in a 12-week, split-body, placebo-comparison study.4 Treated sites included trunk, upper and lower extremities, elbows, knees, palms and soles. An investigator-blinded 4-week study of BMV foam for moderate to severe scalp psoriasis (n = 79) demonstrated significant improvement with both once and twice daily application.5 • CP foam, applied twice daily for 2 weeks, proved to be highly efficacious for non-scalp psoriasis in a double-blind, placebo controlled study of adult patients with involvement of up to 20% body surface area.6 At the end of 2 weeks, 71% in the CP foam group (n = 139 ) and 22% in the placebo foam vehicle group (n = 140) demonstrated complete or almost complete clearance of disease by physician global assessment. At follow-up 2 weeks after completion of therapy, efficacy was sustained in 57% of patients treated with CP foam compared to 17% in the placebo-treated arm. Patient assessment of response proved to be comparable to the assessments completed by the study investigators. No significant local or systemic reactions were noted in patients utilizing either active therapy or the placebo foam vehicle. • A study of seborrheic dermatitis of the scalp in African American adults (n = 69) treated with BMV foam confirmed significant efficacy and high patient tolerability.7 Patient Acceptability Clinical studies using the foam vehicle for psoriasis, including application for both scalp and non-scalp sites, have demonstrated high compliance (>90%) and minimal discontinuation of therapy due to product intolerance.1,2,4-7 The chemical composition and rapid dispersal of the inert components of the vehicle allow for a resultant effect on skin that’s neither drying or moisturizing. In a quantitative assessment of patient acceptance comparing specific products in foam, ointment, solution, gel, emollient and cream, parameters such as ease of use, application time, texture (“feel”), smell, perception of absorption into skin, tendency for staining and impact on quality of life were evaluated.8 The foam vehicle exhibited the most positive quality-of-life scores and was found by patients to be easy to apply, less time consuming and with favorable smell, feel and less staining. Overall, foam and solution were preferred over the other vehicles tested. In a study of CP foam for non-scalp psoriasis, based on comparison with other previously used vehicles, 73% and 66% of patients rated the foam superior to other vehicles (ie. ointment, cream, gel) in terms of “ease of use” and “ability to use at any sites”, respectively.6 In African-American adult males and females, an evaluation correlating BMV foam use with hair styling methods revealed a high level of acceptance in all patients using hot combs, synthetic braids and “curling perms,” 95.7% with natural hairstyles and 76% using chemical relaxers.7 Read on for specific clinical cases showing patient satisfaction and product efficacy of these foam vehicles. Case #1: Scalp Psoriasis History V.W. is a 62-year-old Caucasian female with a history of scalp psoriasis of “several years” duration. The patient reported prior use of various over-the-counter (OTC) preparations, especially shampoo preparations containing tar, salicylic acid and zinc pyrithione. Previous prescription usage included trials of mid- and high-potency topical corticosteroids with limited satisfaction. In addition to her complaints of redness, scaling and itching, she also expressed frustration regarding social embarrassment and the poor cosmetic acceptability of prior treatments. She said she desired therapy that “is not greasy in my scalp and hair.” Clinical presentation revealed erythema and marked psoriasiform scaling involving approximately 40% of the scalp area, extending beyond the scalp margin onto the neck and periauricular region (see photo top right). Selected Treatment Regimen• Clobetasol propionate foam 0.05% (Olux) applied to scalp at bedtime • Shampoo in the morning with product of choice • Reapply CP foam (Olux) in the morning after drying scalp • Follow-up in 3 weeks to monitor progress. Initial treatment outcome. Marked clearance of disease was achieved within 3 weeks; complete clearance was achieved by 4 weeks (see photo bottom right). Therapy with CP foam was discontinued once affected areas resolved. No adverse reactions were reported. Maintenance therapy outcome. Disease flares were successfully controlled within 1 week of restarting therapy with CP foam. The patient reported periods of remission of 1 to 2 months between treatment episodes utilizing this approach. No adverse reactions were observed or reported over long-term follow-up. Patient perspective. The patient reported complete satisfaction with both the initial and continued responses to treatment, ease of use and cosmetic acceptability of the CP foam, especially after application in the morning prior to social encounter. At follow-up she stated, “I can wear my hair up finally.” Case #2: Hand Dermatitis History P.G., a 52-year-old Caucasian male bartender/waiter, presented with a 2-month history of eczematous dermatitis of the hands (see photo on page 78 at left). Various OTC emollients provided no improvement. The patient preferred not to use protective gloves because of the social and public interaction related to his type of work. The eruption was characterized by erythema, scaling, fine fissuring and marked pruritus located on the dorsal and volar surfaces of the hands and fingers, consistent with contact dermatitis. He was exposed to multiple contactants (ie. water, juices, foods) and frequent hand washing, so the etiology was felt to be irritant in nature. Although clearing the eruption and getting relief from symptoms were his most immediate concerns, he said he wished he could hide his hands from the public because of embarrassment. He was also concerned about the “feel” of any recommended product as he dislikes “using creams or anything greasy” on his hands. Selected Treatment Regimen • Use of a mild skin cleanser (Cetaphil Liquid Cleanser) • Clobetasol propionate foam 0.05 % (Olux) twice daily • Dimethicone 1% water-in-oil hand protectant (Theraseal) applied after CP foam (Olux) • Follow-up in 2 to 3 weeks Initial treatment outcome. Complete clearance was noted within 2 weeks of use (see photo above right). Therapy with CP foam was discontinued upon resolution of signs and symptoms of dermatitis. No adverse reactions were reported. Maintenance therapy outcome. The dimethicone 1% water-in-oil hand protectant was continued each day to reduce the frequency and severity of disease recurrence and was felt to be very beneficial in preventing recurrence of disease. The combination of aluminum magnesium hydroxide stearate and cyclomethicone with dimethicone 1% in a water-in-oil formulation allows for simulation of the aqueous phase-lipid phase bilayer of the normal skin barrier and greater substantivity of the cyclomethicone component. Intermittent disease flares were successfully controlled within <1 week of restarting therapy with CP foam. No adverse reactions were observed or noted. Patient perspective. The patient described marked satisfaction with both the CP foam and the hand protectant. He found the foam to be non-irritating and non-drying and liked its rapid disappearance into the skin without the “feel” of any residue. Case #3: Diffuse Psoriasis History R.B. is a 64-year-old Caucasian male with a long history of generalized psoriasis involving his trunk and extremities. Prior treatment included limited response to various topical corticosteroids and calcipotriene 0.05% ointment. He had a history of diffuse actinic damage and multiple squamous cell carcinomas, so phototherapy wasn’t a reasonable option. He refused systemic therapy such as retinoids, methotrexate and cyclosporin over concern about potential adverse reactions. Partial response was noted with topical combination therapy with triamcinolone 0.1% cream applied in the a.m. and tazarotene 0.1% gel applied at night. The patient ex-pressed a strong desire for greater control of psoriasis. Selected Treatment Regimen • Betamethasone valerate foam 0.12% (Luxiq) applied each morning • Discontinue triamcinolone 0.1% cream • Continue tazarotene 0.1% gel (Tazorac) each night • Follow-up in 6 weeks Initial treatment outcome. Marked improvement with multiple areas completely cleared. Residual patches with decreased erythema and scaling were noted after 6 weeks of therapy. No adverse reactions were observed. Maintenance therapy outcome. Control of diffuse psoriasis was maintained over the ensuing 2 months with tapering of BMV foam to 3 mornings each week and continued use of tazarotene 0.1% gel each night. After this point, only sporadic localized recurrences of psoriasis were noted and controlled with reinstitution of therapy until clearance was achieved. Patient perspective. The patient noted marked satisfaction with the therapeutic response and the ease of application of both the CP foam and tazarotene gel vehicles to a wide body surface area. Case #4: Acute Contact Dermatitis History A 44-year-old Caucasian male presented with an acute eczematous dermatitis with marked pruritus on the right distal foot (photo, top right). The patient had used a neomycin-containing “triple antibiotic” cream on a small, superficial abrasion 4 to 5 days prior to the onset of the eruption. Selected Treatment Regimen • Mild skin cleanser (Cetaphil Liquid Cleanser) • Discontinuation of triple antibiotic cream • Clobetasol propionate 0.05 % foam (Olux) twice daily x 2 weeks • Follow-up in 2 to 3 weeks Initial treatment outcome. Complete resolution of the eruption and pruritus observed at follow-up (photo, bottom right). CP foam therapy was discontinued without recurrence of the eruption. No adverse effects were noted, including local application-site reactions. Maintenance therapy outcome. Avoidance of over-the counter (OTC) topical antibiotic preparations, including neomycin-containing products, was recommended. The patient subsequently utilized preparations containing bacitracin and polymyxin B on other occasions without recurrence of contact dermatitis. Patient perspective. The patient expressed marked satisfaction with the quick response, stating that the pruritus dramatically decreased within the first 1 to 2 days and the visible “rash” improved significantly “within the first few days.” The foam vehicle easily spread on the skin, including applications early in treatment when the eruption was more acute and exudative. A Good Recommendation The research and cases presented above demonstrate the efficacy of foam vehicles for the application of topical corticosteroids. The high patient acceptance of these foam vehicles makes for excellent compliance and, therefore, greater efficacy.
Using a Foam Vehicle For Dermatologic Applications
T he recent development of a foam vehicle for application of topical corticosteroids has proven beneficial for the treatment of scalp eruptions such as psoriasis and seborrheic dermatitis. Studies have demonstrated enhanced penetration and a high level of cosmetic acceptability as a result of rapid disappearance and penetration of the foam into skin without a residue. In addition to FDA-approved usage on scalp, further experience has shown that the foam vehicle may be used to treat other cutaneous body sites with successful results demonstrated for a wide variety of steroid responsive dermatoses. Here’s a look at the research supporting use of the foam vehicle and various clinical applications through illustrative case studies. Development And Evolution Of The Foam Vehicle Presently, two topical corticosteroids are available in the United States in a specially formulated foam vehicle. Betamethasone valerate 0.12% foam (BMV foam) has been available for use since FDA approval in 1999 and clobetasol propionate 0.05 % foam (CP foam) since approval in 2000. Other compounds also demonstrate stability in the foam vehicle and are currently under development. The foam formulation has been described in the literature as a novel vehicle with enhanced delivery.1,2 Enhanced delivery refers to rapid product dispersal upon application and enhanced percutaneous absorption in human skin models compared to other vehicle types. Prior to contact with skin, the foam remains stable. Upon skin contact, natural body heat causes rapid volatilization of inert contents with deposition of active drug onto the skin surface. In order to facilitate efficient use, it’s recommended that the patient place a small amount of the foam into the cap prior to application (see photo on page 76). As the foam contains <4. 5% solid ingredients, there’s little-to-no detectable residue on skin.1-3 “Quick break” dispersal also allows for easy spreadability, which is especially important when applying the foam to an extensive body surface area or intertriginous site. In studies using human skin, enhanced percutaneous penetration has been demonstrated in vitro with both BMV foam and CP foam. Within the first 24 hours, BMV foam exhibited six-fold greater skin penetration than betamethasone valerate 0.1% lotion (BMV lotion).1 In a comparison of CP foam and clobetasol propionate 0.05% solution (CP solution), CP foam demonstrated statistically significant greater peak and total absorption after both 12 hours and 48 hours.2 Vasoconstrictor assay studies revealed BMV foam as medium potency and CP foam as ultra-high potency. Clinical Efficacy Double-blind, placebo-controlled clinical studies have confirmed the efficacy of both corticosteroid foam formulations for moderate to severe scalp psoriasis in adults. These studies included active drug comparisons using other vehicles. Here’s a look at results from these studies. • Based on investigator global assessment after 28 days of active drug use, patients were judged to be “clear or almost clear” in 72% of patients treated with BMV foam (n = 57) and 47% treated with BMV lotion (n = 58).1 • Similar evaluation comparing 14 days of active treatment with CP foam (n = 62) or CP solution (n = 63) rated 74% of patients as “completely clear or almost clear” in the foam-treated group and 63% in the solution-treated group.2 Signs and symptoms of psoriasis demonstrated greater response after both 1 and 2 weeks of active therapy and at follow-up 2 weeks post-therapy in patients treated with CP foam as compared to CP solution or placebo. The adverse reaction profile of both foam compounds has proven to be favorable in multiple studies, and comparable to placebo and other tested comparator vehicles. • The efficacy of BMV foam for non-scalp psoriasis (n = 37) was confirmed in a 12-week, split-body, placebo-comparison study.4 Treated sites included trunk, upper and lower extremities, elbows, knees, palms and soles. An investigator-blinded 4-week study of BMV foam for moderate to severe scalp psoriasis (n = 79) demonstrated significant improvement with both once and twice daily application.5 • CP foam, applied twice daily for 2 weeks, proved to be highly efficacious for non-scalp psoriasis in a double-blind, placebo controlled study of adult patients with involvement of up to 20% body surface area.6 At the end of 2 weeks, 71% in the CP foam group (n = 139 ) and 22% in the placebo foam vehicle group (n = 140) demonstrated complete or almost complete clearance of disease by physician global assessment. At follow-up 2 weeks after completion of therapy, efficacy was sustained in 57% of patients treated with CP foam compared to 17% in the placebo-treated arm. Patient assessment of response proved to be comparable to the assessments completed by the study investigators. No significant local or systemic reactions were noted in patients utilizing either active therapy or the placebo foam vehicle. • A study of seborrheic dermatitis of the scalp in African American adults (n = 69) treated with BMV foam confirmed significant efficacy and high patient tolerability.7 Patient Acceptability Clinical studies using the foam vehicle for psoriasis, including application for both scalp and non-scalp sites, have demonstrated high compliance (>90%) and minimal discontinuation of therapy due to product intolerance.1,2,4-7 The chemical composition and rapid dispersal of the inert components of the vehicle allow for a resultant effect on skin that’s neither drying or moisturizing. In a quantitative assessment of patient acceptance comparing specific products in foam, ointment, solution, gel, emollient and cream, parameters such as ease of use, application time, texture (“feel”), smell, perception of absorption into skin, tendency for staining and impact on quality of life were evaluated.8 The foam vehicle exhibited the most positive quality-of-life scores and was found by patients to be easy to apply, less time consuming and with favorable smell, feel and less staining. Overall, foam and solution were preferred over the other vehicles tested. In a study of CP foam for non-scalp psoriasis, based on comparison with other previously used vehicles, 73% and 66% of patients rated the foam superior to other vehicles (ie. ointment, cream, gel) in terms of “ease of use” and “ability to use at any sites”, respectively.6 In African-American adult males and females, an evaluation correlating BMV foam use with hair styling methods revealed a high level of acceptance in all patients using hot combs, synthetic braids and “curling perms,” 95.7% with natural hairstyles and 76% using chemical relaxers.7 Read on for specific clinical cases showing patient satisfaction and product efficacy of these foam vehicles. Case #1: Scalp Psoriasis History V.W. is a 62-year-old Caucasian female with a history of scalp psoriasis of “several years” duration. The patient reported prior use of various over-the-counter (OTC) preparations, especially shampoo preparations containing tar, salicylic acid and zinc pyrithione. Previous prescription usage included trials of mid- and high-potency topical corticosteroids with limited satisfaction. In addition to her complaints of redness, scaling and itching, she also expressed frustration regarding social embarrassment and the poor cosmetic acceptability of prior treatments. She said she desired therapy that “is not greasy in my scalp and hair.” Clinical presentation revealed erythema and marked psoriasiform scaling involving approximately 40% of the scalp area, extending beyond the scalp margin onto the neck and periauricular region (see photo top right). Selected Treatment Regimen• Clobetasol propionate foam 0.05% (Olux) applied to scalp at bedtime • Shampoo in the morning with product of choice • Reapply CP foam (Olux) in the morning after drying scalp • Follow-up in 3 weeks to monitor progress. Initial treatment outcome. Marked clearance of disease was achieved within 3 weeks; complete clearance was achieved by 4 weeks (see photo bottom right). Therapy with CP foam was discontinued once affected areas resolved. No adverse reactions were reported. Maintenance therapy outcome. Disease flares were successfully controlled within 1 week of restarting therapy with CP foam. The patient reported periods of remission of 1 to 2 months between treatment episodes utilizing this approach. No adverse reactions were observed or reported over long-term follow-up. Patient perspective. The patient reported complete satisfaction with both the initial and continued responses to treatment, ease of use and cosmetic acceptability of the CP foam, especially after application in the morning prior to social encounter. At follow-up she stated, “I can wear my hair up finally.” Case #2: Hand Dermatitis History P.G., a 52-year-old Caucasian male bartender/waiter, presented with a 2-month history of eczematous dermatitis of the hands (see photo on page 78 at left). Various OTC emollients provided no improvement. The patient preferred not to use protective gloves because of the social and public interaction related to his type of work. The eruption was characterized by erythema, scaling, fine fissuring and marked pruritus located on the dorsal and volar surfaces of the hands and fingers, consistent with contact dermatitis. He was exposed to multiple contactants (ie. water, juices, foods) and frequent hand washing, so the etiology was felt to be irritant in nature. Although clearing the eruption and getting relief from symptoms were his most immediate concerns, he said he wished he could hide his hands from the public because of embarrassment. He was also concerned about the “feel” of any recommended product as he dislikes “using creams or anything greasy” on his hands. Selected Treatment Regimen • Use of a mild skin cleanser (Cetaphil Liquid Cleanser) • Clobetasol propionate foam 0.05 % (Olux) twice daily • Dimethicone 1% water-in-oil hand protectant (Theraseal) applied after CP foam (Olux) • Follow-up in 2 to 3 weeks Initial treatment outcome. Complete clearance was noted within 2 weeks of use (see photo above right). Therapy with CP foam was discontinued upon resolution of signs and symptoms of dermatitis. No adverse reactions were reported. Maintenance therapy outcome. The dimethicone 1% water-in-oil hand protectant was continued each day to reduce the frequency and severity of disease recurrence and was felt to be very beneficial in preventing recurrence of disease. The combination of aluminum magnesium hydroxide stearate and cyclomethicone with dimethicone 1% in a water-in-oil formulation allows for simulation of the aqueous phase-lipid phase bilayer of the normal skin barrier and greater substantivity of the cyclomethicone component. Intermittent disease flares were successfully controlled within <1 week of restarting therapy with CP foam. No adverse reactions were observed or noted. Patient perspective. The patient described marked satisfaction with both the CP foam and the hand protectant. He found the foam to be non-irritating and non-drying and liked its rapid disappearance into the skin without the “feel” of any residue. Case #3: Diffuse Psoriasis History R.B. is a 64-year-old Caucasian male with a long history of generalized psoriasis involving his trunk and extremities. Prior treatment included limited response to various topical corticosteroids and calcipotriene 0.05% ointment. He had a history of diffuse actinic damage and multiple squamous cell carcinomas, so phototherapy wasn’t a reasonable option. He refused systemic therapy such as retinoids, methotrexate and cyclosporin over concern about potential adverse reactions. Partial response was noted with topical combination therapy with triamcinolone 0.1% cream applied in the a.m. and tazarotene 0.1% gel applied at night. The patient ex-pressed a strong desire for greater control of psoriasis. Selected Treatment Regimen • Betamethasone valerate foam 0.12% (Luxiq) applied each morning • Discontinue triamcinolone 0.1% cream • Continue tazarotene 0.1% gel (Tazorac) each night • Follow-up in 6 weeks Initial treatment outcome. Marked improvement with multiple areas completely cleared. Residual patches with decreased erythema and scaling were noted after 6 weeks of therapy. No adverse reactions were observed. Maintenance therapy outcome. Control of diffuse psoriasis was maintained over the ensuing 2 months with tapering of BMV foam to 3 mornings each week and continued use of tazarotene 0.1% gel each night. After this point, only sporadic localized recurrences of psoriasis were noted and controlled with reinstitution of therapy until clearance was achieved. Patient perspective. The patient noted marked satisfaction with the therapeutic response and the ease of application of both the CP foam and tazarotene gel vehicles to a wide body surface area. Case #4: Acute Contact Dermatitis History A 44-year-old Caucasian male presented with an acute eczematous dermatitis with marked pruritus on the right distal foot (photo, top right). The patient had used a neomycin-containing “triple antibiotic” cream on a small, superficial abrasion 4 to 5 days prior to the onset of the eruption. Selected Treatment Regimen • Mild skin cleanser (Cetaphil Liquid Cleanser) • Discontinuation of triple antibiotic cream • Clobetasol propionate 0.05 % foam (Olux) twice daily x 2 weeks • Follow-up in 2 to 3 weeks Initial treatment outcome. Complete resolution of the eruption and pruritus observed at follow-up (photo, bottom right). CP foam therapy was discontinued without recurrence of the eruption. No adverse effects were noted, including local application-site reactions. Maintenance therapy outcome. Avoidance of over-the counter (OTC) topical antibiotic preparations, including neomycin-containing products, was recommended. The patient subsequently utilized preparations containing bacitracin and polymyxin B on other occasions without recurrence of contact dermatitis. Patient perspective. The patient expressed marked satisfaction with the quick response, stating that the pruritus dramatically decreased within the first 1 to 2 days and the visible “rash” improved significantly “within the first few days.” The foam vehicle easily spread on the skin, including applications early in treatment when the eruption was more acute and exudative. A Good Recommendation The research and cases presented above demonstrate the efficacy of foam vehicles for the application of topical corticosteroids. The high patient acceptance of these foam vehicles makes for excellent compliance and, therefore, greater efficacy.
T he recent development of a foam vehicle for application of topical corticosteroids has proven beneficial for the treatment of scalp eruptions such as psoriasis and seborrheic dermatitis. Studies have demonstrated enhanced penetration and a high level of cosmetic acceptability as a result of rapid disappearance and penetration of the foam into skin without a residue. In addition to FDA-approved usage on scalp, further experience has shown that the foam vehicle may be used to treat other cutaneous body sites with successful results demonstrated for a wide variety of steroid responsive dermatoses. Here’s a look at the research supporting use of the foam vehicle and various clinical applications through illustrative case studies. Development And Evolution Of The Foam Vehicle Presently, two topical corticosteroids are available in the United States in a specially formulated foam vehicle. Betamethasone valerate 0.12% foam (BMV foam) has been available for use since FDA approval in 1999 and clobetasol propionate 0.05 % foam (CP foam) since approval in 2000. Other compounds also demonstrate stability in the foam vehicle and are currently under development. The foam formulation has been described in the literature as a novel vehicle with enhanced delivery.1,2 Enhanced delivery refers to rapid product dispersal upon application and enhanced percutaneous absorption in human skin models compared to other vehicle types. Prior to contact with skin, the foam remains stable. Upon skin contact, natural body heat causes rapid volatilization of inert contents with deposition of active drug onto the skin surface. In order to facilitate efficient use, it’s recommended that the patient place a small amount of the foam into the cap prior to application (see photo on page 76). As the foam contains <4. 5% solid ingredients, there’s little-to-no detectable residue on skin.1-3 “Quick break” dispersal also allows for easy spreadability, which is especially important when applying the foam to an extensive body surface area or intertriginous site. In studies using human skin, enhanced percutaneous penetration has been demonstrated in vitro with both BMV foam and CP foam. Within the first 24 hours, BMV foam exhibited six-fold greater skin penetration than betamethasone valerate 0.1% lotion (BMV lotion).1 In a comparison of CP foam and clobetasol propionate 0.05% solution (CP solution), CP foam demonstrated statistically significant greater peak and total absorption after both 12 hours and 48 hours.2 Vasoconstrictor assay studies revealed BMV foam as medium potency and CP foam as ultra-high potency. Clinical Efficacy Double-blind, placebo-controlled clinical studies have confirmed the efficacy of both corticosteroid foam formulations for moderate to severe scalp psoriasis in adults. These studies included active drug comparisons using other vehicles. Here’s a look at results from these studies. • Based on investigator global assessment after 28 days of active drug use, patients were judged to be “clear or almost clear” in 72% of patients treated with BMV foam (n = 57) and 47% treated with BMV lotion (n = 58).1 • Similar evaluation comparing 14 days of active treatment with CP foam (n = 62) or CP solution (n = 63) rated 74% of patients as “completely clear or almost clear” in the foam-treated group and 63% in the solution-treated group.2 Signs and symptoms of psoriasis demonstrated greater response after both 1 and 2 weeks of active therapy and at follow-up 2 weeks post-therapy in patients treated with CP foam as compared to CP solution or placebo. The adverse reaction profile of both foam compounds has proven to be favorable in multiple studies, and comparable to placebo and other tested comparator vehicles. • The efficacy of BMV foam for non-scalp psoriasis (n = 37) was confirmed in a 12-week, split-body, placebo-comparison study.4 Treated sites included trunk, upper and lower extremities, elbows, knees, palms and soles. An investigator-blinded 4-week study of BMV foam for moderate to severe scalp psoriasis (n = 79) demonstrated significant improvement with both once and twice daily application.5 • CP foam, applied twice daily for 2 weeks, proved to be highly efficacious for non-scalp psoriasis in a double-blind, placebo controlled study of adult patients with involvement of up to 20% body surface area.6 At the end of 2 weeks, 71% in the CP foam group (n = 139 ) and 22% in the placebo foam vehicle group (n = 140) demonstrated complete or almost complete clearance of disease by physician global assessment. At follow-up 2 weeks after completion of therapy, efficacy was sustained in 57% of patients treated with CP foam compared to 17% in the placebo-treated arm. Patient assessment of response proved to be comparable to the assessments completed by the study investigators. No significant local or systemic reactions were noted in patients utilizing either active therapy or the placebo foam vehicle. • A study of seborrheic dermatitis of the scalp in African American adults (n = 69) treated with BMV foam confirmed significant efficacy and high patient tolerability.7 Patient Acceptability Clinical studies using the foam vehicle for psoriasis, including application for both scalp and non-scalp sites, have demonstrated high compliance (>90%) and minimal discontinuation of therapy due to product intolerance.1,2,4-7 The chemical composition and rapid dispersal of the inert components of the vehicle allow for a resultant effect on skin that’s neither drying or moisturizing. In a quantitative assessment of patient acceptance comparing specific products in foam, ointment, solution, gel, emollient and cream, parameters such as ease of use, application time, texture (“feel”), smell, perception of absorption into skin, tendency for staining and impact on quality of life were evaluated.8 The foam vehicle exhibited the most positive quality-of-life scores and was found by patients to be easy to apply, less time consuming and with favorable smell, feel and less staining. Overall, foam and solution were preferred over the other vehicles tested. In a study of CP foam for non-scalp psoriasis, based on comparison with other previously used vehicles, 73% and 66% of patients rated the foam superior to other vehicles (ie. ointment, cream, gel) in terms of “ease of use” and “ability to use at any sites”, respectively.6 In African-American adult males and females, an evaluation correlating BMV foam use with hair styling methods revealed a high level of acceptance in all patients using hot combs, synthetic braids and “curling perms,” 95.7% with natural hairstyles and 76% using chemical relaxers.7 Read on for specific clinical cases showing patient satisfaction and product efficacy of these foam vehicles. Case #1: Scalp Psoriasis History V.W. is a 62-year-old Caucasian female with a history of scalp psoriasis of “several years” duration. The patient reported prior use of various over-the-counter (OTC) preparations, especially shampoo preparations containing tar, salicylic acid and zinc pyrithione. Previous prescription usage included trials of mid- and high-potency topical corticosteroids with limited satisfaction. In addition to her complaints of redness, scaling and itching, she also expressed frustration regarding social embarrassment and the poor cosmetic acceptability of prior treatments. She said she desired therapy that “is not greasy in my scalp and hair.” Clinical presentation revealed erythema and marked psoriasiform scaling involving approximately 40% of the scalp area, extending beyond the scalp margin onto the neck and periauricular region (see photo top right). Selected Treatment Regimen• Clobetasol propionate foam 0.05% (Olux) applied to scalp at bedtime • Shampoo in the morning with product of choice • Reapply CP foam (Olux) in the morning after drying scalp • Follow-up in 3 weeks to monitor progress. Initial treatment outcome. Marked clearance of disease was achieved within 3 weeks; complete clearance was achieved by 4 weeks (see photo bottom right). Therapy with CP foam was discontinued once affected areas resolved. No adverse reactions were reported. Maintenance therapy outcome. Disease flares were successfully controlled within 1 week of restarting therapy with CP foam. The patient reported periods of remission of 1 to 2 months between treatment episodes utilizing this approach. No adverse reactions were observed or reported over long-term follow-up. Patient perspective. The patient reported complete satisfaction with both the initial and continued responses to treatment, ease of use and cosmetic acceptability of the CP foam, especially after application in the morning prior to social encounter. At follow-up she stated, “I can wear my hair up finally.” Case #2: Hand Dermatitis History P.G., a 52-year-old Caucasian male bartender/waiter, presented with a 2-month history of eczematous dermatitis of the hands (see photo on page 78 at left). Various OTC emollients provided no improvement. The patient preferred not to use protective gloves because of the social and public interaction related to his type of work. The eruption was characterized by erythema, scaling, fine fissuring and marked pruritus located on the dorsal and volar surfaces of the hands and fingers, consistent with contact dermatitis. He was exposed to multiple contactants (ie. water, juices, foods) and frequent hand washing, so the etiology was felt to be irritant in nature. Although clearing the eruption and getting relief from symptoms were his most immediate concerns, he said he wished he could hide his hands from the public because of embarrassment. He was also concerned about the “feel” of any recommended product as he dislikes “using creams or anything greasy” on his hands. Selected Treatment Regimen • Use of a mild skin cleanser (Cetaphil Liquid Cleanser) • Clobetasol propionate foam 0.05 % (Olux) twice daily • Dimethicone 1% water-in-oil hand protectant (Theraseal) applied after CP foam (Olux) • Follow-up in 2 to 3 weeks Initial treatment outcome. Complete clearance was noted within 2 weeks of use (see photo above right). Therapy with CP foam was discontinued upon resolution of signs and symptoms of dermatitis. No adverse reactions were reported. Maintenance therapy outcome. The dimethicone 1% water-in-oil hand protectant was continued each day to reduce the frequency and severity of disease recurrence and was felt to be very beneficial in preventing recurrence of disease. The combination of aluminum magnesium hydroxide stearate and cyclomethicone with dimethicone 1% in a water-in-oil formulation allows for simulation of the aqueous phase-lipid phase bilayer of the normal skin barrier and greater substantivity of the cyclomethicone component. Intermittent disease flares were successfully controlled within <1 week of restarting therapy with CP foam. No adverse reactions were observed or noted. Patient perspective. The patient described marked satisfaction with both the CP foam and the hand protectant. He found the foam to be non-irritating and non-drying and liked its rapid disappearance into the skin without the “feel” of any residue. Case #3: Diffuse Psoriasis History R.B. is a 64-year-old Caucasian male with a long history of generalized psoriasis involving his trunk and extremities. Prior treatment included limited response to various topical corticosteroids and calcipotriene 0.05% ointment. He had a history of diffuse actinic damage and multiple squamous cell carcinomas, so phototherapy wasn’t a reasonable option. He refused systemic therapy such as retinoids, methotrexate and cyclosporin over concern about potential adverse reactions. Partial response was noted with topical combination therapy with triamcinolone 0.1% cream applied in the a.m. and tazarotene 0.1% gel applied at night. The patient ex-pressed a strong desire for greater control of psoriasis. Selected Treatment Regimen • Betamethasone valerate foam 0.12% (Luxiq) applied each morning • Discontinue triamcinolone 0.1% cream • Continue tazarotene 0.1% gel (Tazorac) each night • Follow-up in 6 weeks Initial treatment outcome. Marked improvement with multiple areas completely cleared. Residual patches with decreased erythema and scaling were noted after 6 weeks of therapy. No adverse reactions were observed. Maintenance therapy outcome. Control of diffuse psoriasis was maintained over the ensuing 2 months with tapering of BMV foam to 3 mornings each week and continued use of tazarotene 0.1% gel each night. After this point, only sporadic localized recurrences of psoriasis were noted and controlled with reinstitution of therapy until clearance was achieved. Patient perspective. The patient noted marked satisfaction with the therapeutic response and the ease of application of both the CP foam and tazarotene gel vehicles to a wide body surface area. Case #4: Acute Contact Dermatitis History A 44-year-old Caucasian male presented with an acute eczematous dermatitis with marked pruritus on the right distal foot (photo, top right). The patient had used a neomycin-containing “triple antibiotic” cream on a small, superficial abrasion 4 to 5 days prior to the onset of the eruption. Selected Treatment Regimen • Mild skin cleanser (Cetaphil Liquid Cleanser) • Discontinuation of triple antibiotic cream • Clobetasol propionate 0.05 % foam (Olux) twice daily x 2 weeks • Follow-up in 2 to 3 weeks Initial treatment outcome. Complete resolution of the eruption and pruritus observed at follow-up (photo, bottom right). CP foam therapy was discontinued without recurrence of the eruption. No adverse effects were noted, including local application-site reactions. Maintenance therapy outcome. Avoidance of over-the counter (OTC) topical antibiotic preparations, including neomycin-containing products, was recommended. The patient subsequently utilized preparations containing bacitracin and polymyxin B on other occasions without recurrence of contact dermatitis. Patient perspective. The patient expressed marked satisfaction with the quick response, stating that the pruritus dramatically decreased within the first 1 to 2 days and the visible “rash” improved significantly “within the first few days.” The foam vehicle easily spread on the skin, including applications early in treatment when the eruption was more acute and exudative. A Good Recommendation The research and cases presented above demonstrate the efficacy of foam vehicles for the application of topical corticosteroids. The high patient acceptance of these foam vehicles makes for excellent compliance and, therefore, greater efficacy.